 
Here the reagent is a constant direct electrical current of known magnitude that consumes the sample. This involves measuring mass of reagent that reacts completely with the sample. Indicators are added to the solution mixtures to produce an observable physical change at the end-point or near equivalence point ii. Best Answer Copy Square Planar, Like XeF4 Wiki User 02:58:45 This answer is: Study guides Science 17 cards Is glucose solution a homogenous mixture Properties that describe the. The two values are usually different and the difference gives the titration error. Chemistry: The Molecular Science 5th Edition ISBN: 9781285199047 Author: John W. Predict the shape of each molecule, and explain why the shapes differ. Equivalence point is a point in titration when the amount of standard solution added is exactly equal to the amount of the sample whereas end-point is the point in titration when a physical change occurs that is associated with a condition of chemical equivalence. The molecules SiF4, SF4, and XeF4 have molecular formulas of the type AF4, but the molecules have different molecular geometries. Equivalence point and end-point are confused to mean the same but they are totally different. Back titration is the process by which the excess of the standard solution used to consume the sample is determined by titration with a second standard solution. Titration is the process in which the standard reagent is added to a solution of the sample until the reaction is judged to be complete. In solid form, the XeF4 has a density of 4.040 g cm3 and has a solid white look. It is a noble gas with the chemical formula Xe + 2F2 -> XeF4. It is the first binary chemical found in the world. 6 7 The structure is square planar, as has been confirmed by neutron diffraction studies. The chemical compound XeF4 (Xenon Tetrafluoride) comprises Xenon and Fluoride atoms. Its structure was determined by both NMR spectroscopy and X-ray crystallography in 1963. The reagent of exactly known concentration is refered to as standard reagent. Xenon tetrafluoride is a colorless crystalline solid that sublimes at 117 C. 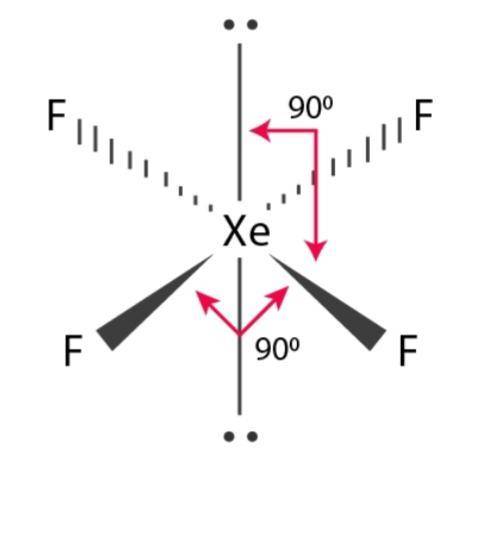
This involves measuring volume of solution of known concentration that is needed to react completely with the sample. Titrimetric methods include powerful group of quantitative procedures that are based on measuring the amount of reagent consumed by the analyte. The volume or mass of the reagent needed to react completely with a fixed quantity of the analyte is obtained from which the amount of analyte is determined. The two groups on the left and the six on the right are the main. Titration is based on a reaction between the analyte (unknown sample) and the regent of known concentration and reaction stoichiometry. Elements are organized into 18 vertical columns, or groups, and 7 horizontal rows, or periods. TITRIMETRIC MTHODS Titrimetric methods are widely used in chemistry to determine oxidants, reductants, acids, bases, metal ions, etc. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
